ELUVIA™ DRUG-ELUTING VASCULAR STENT SYSTEM DEMONSTRATES 12-MONTH PRIMARY PATENCY OF 96.1 PERCENT
MAJESTIC Trial Data Support Strong Safety Profile with Low Target Lesion Revascularization Rate
Video
Comments from Prof. Stefan Müller-Hülsbeck, M.D., PhD
Video animation
Photos
CONTACTS
Media:
Trish Backes
Global Media Relations
Boston Scientific Corporation
651-582-5887 (office)
[email protected]
Simonetta Balbi
39 3387936422 (mobile)
39 0106060281 (direct)
European Media Relations
Boston Scientific Corporation
[email protected]
Investors:
Susie Lisa, CFA
Investor Relations
Boston Scientific Corporation
(508) 683-5565 (office)
[email protected]
Related Links
Follow Us On Twitter
PR Newswire – MARLBOROUGH, Mass., Sept. 28, 2015 New 12-month clinical trial outcomes assessing the safety and performance of the Boston Scientific (NYSE: BSX) Eluvia™ Drug-Eluting Vascular Stent System reflect a primary patency rate1 of more than 96 percent. These results represent the highest 12-month primary patency reported for an interventional treatment of femoropopliteal artery lesions among comparable trials. The Eluvia Stent System is an advanced treatment option for patients with narrowing or blockages in the superficial femoral artery (SFA) or proximal popliteal artery (PPA), a result of peripheral artery disease (PAD).
Results from the MAJESTIC trial, which were presented at the Cardiovascular and Interventional Radiological Society of Europe (CIRSE) annual meeting in Lisbon, Portugal, also included a low 12-month target lesion revascularization (TLR) rate of 3.8 percent, with no observed stent fractures and no amputations.
“Achieving a 96 percent primary patency rate at one year, with low major adverse events, is exceptional,” said Professor Stefan Müller-Hülsbeck, M.D., PhD, principal investigator and chairman, Vascular Center Diako Flensburg and head of the Department of Diagnostic and Interventional Radiology /Neuroradiology, Academic Hospitals Flensburg, Germany. “In my opinion, the sustained release of paclitaxel enabled by this technology could represent a significant advancement in the treatment of patients with peripheral arterial disease.”
The MAJESTIC trial, a prospective, multicenter clinical trial, enrolled 57 patients across Europe, Australia and New Zealand, with an average lesion length of 70.8mm. The trial included a high percentage of complex lesions, with 46% of lesions classified as total occlusions and 65% identified as severely calcified.
Data from the MAJESTIC trial are expected to support global regulatory submissions. In addition, Boston Scientific has received an Investigational Device Exemption (IDE) from the Food and Drug Administration (FDA) to begin a global, pivotal study of the Eluvia Stent System. The study, named the IMPERIAL trial, is designed to support future regulatory submissions and patient enrollment is expected to begin in the coming months.
“The MAJESTIC trial demonstrates our commitment to bringing forward innovative therapies that can make a difference in the lives of patients with PAD who face risks of serious complications including infections and amputations,” said Jeff Mirviss, senior vice president and president, Peripheral Interventions, Boston Scientific. “The Eluvia Stent is built on a foundation of more than 15 years of design, development and clinical leadership in drug-eluting technologies at Boston Scientific.”
About the Eluvia Stent System
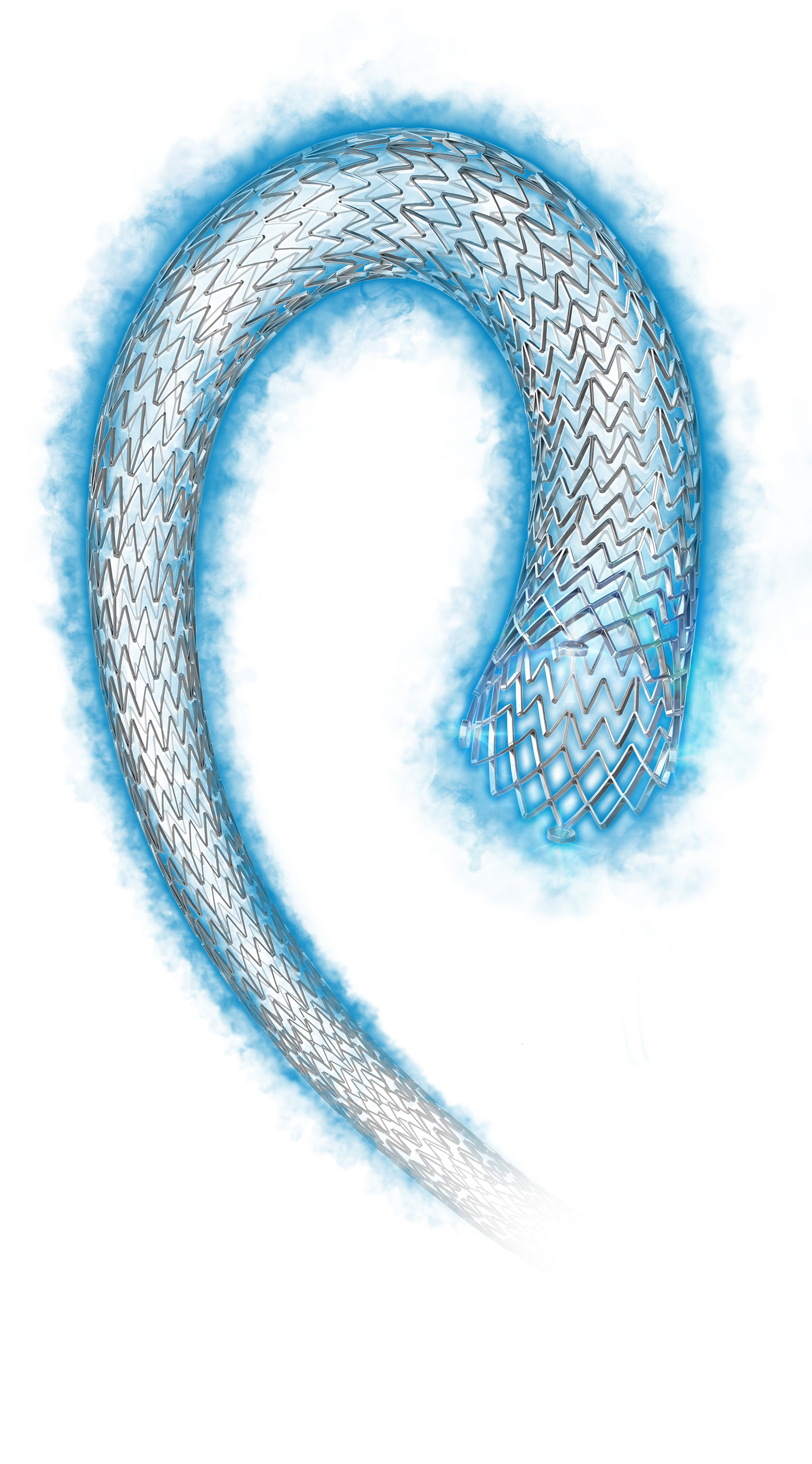
The Eluvia Stent System is the first stent specifically designed for deployment in the superficial femoral artery (SFA) that utilizes the anti-restenotic drug paclitaxel in conjunction with a polymer. This drug and polymer combination is intended to facilitate sustained release of the drug over the period of time when restenosis is most likely to occur, preventing tissue growth that might otherwise block the stented artery. The Eluvia Stent System is built on the Innova™ Stent System platform, consisting of a self-expanding nitinol stent and an advanced, 6F low-profile triaxial delivery system for added support and placement accuracy. The innovative stent architecture features a closed-cell design at each end of the stent for more predictable deployment, and an open-cell design along the stent body for improved flexibility, strength and fracture resistance. View or download an image of the Eluvia Stent System.
The Eluvia Stent System is pending CE Mark approval. In the U.S., the Eluvia Stent System is an investigational device and is not available for sale.
About Peripheral Artery Disease
Peripheral artery disease is a circulatory disorder that results from a build-up of plaque in one or more of the arteries, most often in the legs. As the disease progresses, plaque accumulation may significantly reduce blood flow through the arteries, resulting in pain and increasing disability, with severe cases often leading to amputation of the affected limb. It is estimated that 200 million people are affected by PAD worldwide.
About Boston Scientific
Boston Scientific transforms lives through innovative medical solutions that improve the health of patients around the world. As a global medical technology leader for more than 35 years, we advance science for life by providing a broad range of high performance solutions that address unmet patient needs and reduce the cost of healthcare. For more information, visit www.bostonscientific.com and connect on Twitter and Facebook.
1. Primary patency defined as duplex ultrasound peak systolic velocity ratio ≤2.5 and absence of TLR or bypass; data reflect actual values (not Kaplan Meier estimates).
Cautionary Statement Regarding Forward-Looking Statements
This press release contains forward-looking statements within the meaning of Section 27A of the Securities Act of 1933 and Section 21E of the Securities Exchange Act of 1934. Forward-looking statements may be identified by words like “anticipate,” “expect,” “project,” “believe,” “plan,” “estimate,” “intend” and similar words. These forward-looking statements are based on our beliefs, assumptions and estimates using information available to us at the time and are not intended to be guarantees of future events or performance. These forward-looking statements include, among other things, statements regarding clinical trials and impact of data and product performance. If our underlying assumptions turn out to be incorrect, or if certain risks or uncertainties materialize, actual results could vary materially from the expectations and projections expressed or implied by our forward-looking statements. These factors, in some cases, have affected and in the future (together with other factors) could affect our ability to implement our business strategy and may cause actual results to differ materially from those contemplated by the statements expressed in this press release. As a result, readers are cautioned not to place undue reliance on any of our forward-looking statements.
Factors that may cause such differences include, among other things: future economic, competitive, reimbursement and regulatory conditions; new product introductions; demographic trends; intellectual property; litigation; financial market conditions; and future business decisions made by us and our competitors. All of these factors are difficult or impossible to predict accurately and many of them are beyond our control. For a further list and description of these and other important risks and uncertainties that may affect our future operations, see Part I, Item 1A – Risk Factors in our most recent Annual Report on Form 10-K filed with the Securities and Exchange Commission, which we may update in Part II, Item 1A – Risk Factors in Quarterly Reports on Form 10-Q we have filed or will file hereafter. We disclaim any intention or obligation to publicly update or revise any forward-looking statements to reflect any change in our expectations or in events, conditions or circumstances on which those expectations may be based, or that may affect the likelihood that actual results will differ from those contained in the forward-looking statements. This cautionary statement is applicable to all forward-looking statements contained in this document.